
Using a Thermometer
Animating the Experiment
Mixing Hot and Cold Water
Temperature Changes in a Line Graph
The Energy Transfer Experiment
Screen Capture 1
Diagramming the Experiment
Screen Capture 2
Topic: Mixing Hot and Cold Water

Learning Objectives:
The students will find the results of what happens to the temperature of water if an equal amount
of hot and cold water is mixed.
Previous Lessons: The students have learned how to use and read a mercury thermometer. They have learned how temperature is measure (celcius and farenheit).
Materials:
thermometers, 2 film cans, I water flask, science notebook, pens, paper towels
Vocabulary: quantity, hypothesis, evidence
Motivation: Show the class the tools to be used in the experiment. Elicit hypotheses from the students: what will be the evidence of energy transfer (temperature changes: energy transferred from the hot to the cold water, the colored alcohol will expand/contract)?
Procedure:
-Motivation
-Write a hypothesis to the learning objective (sample hypothesis: I think that the water will get warmer.");
-Hand out two film cans to each student;
-Pour hot water into one film can and cold water into the other film can; repeat this process for each student;
-The students will take the temperature of the can of cold water and the can of hot water; they will record the temperatures in their science notebooks;
-The students will mix equal quantities of hot and cold water into a single container;
-The students will measure the temperature and record the changes in their science notebooks;
-The students will hypothesize about mixing unequal quantities of hot and cold water; (sample hypothesis below)
-The students will mix the unequal quantities of hot and cold water and record the results in their science notebooks;
-Discuss the pattern seen among the class' results (regarding the temperature of the mixtures, its
average temperature, its middle temperature);
-Discuss the variables which affect the temperature of the mixture (the hot water might have
cooled a little by the time it was handed out to their table, some of the water might have spilled,
some of the thermometers might be broken).

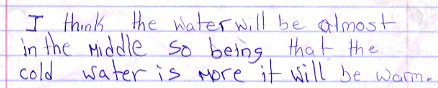

Follow-up:
The students will experiment with energy transfer. They will put a hot water film can inside a
cold water film can and record the energy transfer from one to the other.
Standards:
-- The student produces evidence that demonstrates understanding of Light, heat, electricity,
and magnetism, such as the variation of heat and temperature; how light travels in a straight line
until it strikes an object or how electrical circuits work.
-- The student produces evidence that demonstrates understanding of Big ideas and unifying
concepts, such as order and organization; models, form and function; change and constancy; and
cause and effect.
-- The student uses technology and tools (such as rulers, computers, balances, thermometers,
watches, magnifiers, and microscopes) to gather data and extend the senses.
-- The student collects and analyzes data using concepts and techniques in Mathematics
Standard 4, such as average, data displays, graphing, variability, and sampling.
-- The student represents data and results in multiple ways, such as numbers, tables, and
graphs; drawings, diagrams, and artwork; and technical and creative writing.
-- The student uses facts to support conclusions.
-- The student critiques written and oral explanations, and uses data to resolve disagreements.